Introduction
Outcomes for children with acute myeloid leukemia (AML) remain poor, with ~40% dying from refractory leukemia or treatment-related toxicity (Gamis et al. 2014). NRAS, KRAS, and NF1 mutations occur in over 40% of pediatric AMLs (Bolouri et al. 2018), but efforts to therapeutically target the RAS/mitogen activated protein kinase (MAPK) pathway have been largely unsuccessful. Belvarafenib, a novel type II pan-RAF kinase inhibitor that inhibits mutant monomeric BRAF proteins and activated RAF homo- and heterodimers, has shown promising safety/efficacy data in adult solid cancers (Yen et al. 2021). Here, we investigate belvarafenib in preclinical models of AML, both alone and in combination with the allosteric MEK inhibitor, cobimetinib.
Methods
We used a panel of NRAS (OCI-AML3, HL-60, THP-1) or KRAS (NOMO-1, NB4, and SKM-1) mutant human AML cell lines. Viability was determined by CellTiter-Glo. Synergy was assessed by Bliss Independence and Chou Talalay methods. Transcriptome and proteomic profiling were performed as previously described (Pucciarelli et al. 2020). Mouse AMLs were generated using retroviral insertional mutagenesis (Li et al. 2011). Cryopreserved primary AML cells were injected intravenously into sublethally irradiated recipients that were then treated daily with vehicle, belvarafenib, cobimetinib, or the combination until disease progression. Survival curves were generated using Kaplan-Meier analysis.
Results
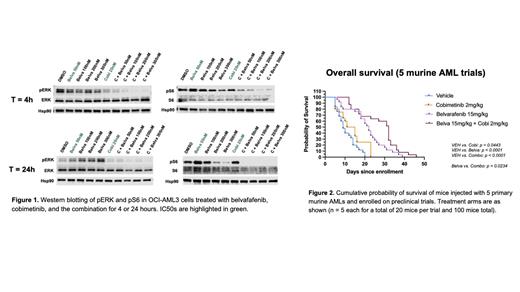
Belvarafenib inhibited the growth of AML cells lines with nanomolar potency at IC50 values ranging from 48nM (OCI-AML3) to 310nM (SKM-1). In all cell lines, belvarafenib and cobimetinib were highly synergistic. Western blotting of OCI-AML3 cells treated with either belvarafenib or cobimetinib at their respective IC50 values (50nM belvarafenib, 20nM cobimetinib) for 4 or 24 hours demonstrated discordant effects on downstream MAPK effector proteins (Fig. 1). Whereas cobimetinib potently suppressed phosphorylated ERK (pERK), belvarafenib unexpectedly had no effect. Phosphorylated S6 (pS6) levels were not reduced by either belvarafenib or cobimetinib at their respective IC50 values, but were dramatically reduced by the drug combination.
To further investigate these unanticipated biochemical findings, we treated OCI-AML3 cells with belvarafenib, cobimetinib or the combination and performed kinome profiling and transcriptome sequencing (RNAseq) analysis. Exposure to 10nM cobimetinib inhibited several kinases at 4 hours with rebound activation of kinases involved in cell cycle progression at 24 hours. Cells treated with 100nM belvarafenib displayed down-regulation of multiple kinases that persisted at 24 hours. Cells treated with the combination at these doses exhibited the most potent inhibition of many kinases; this effect recapitulated the effect of high dose (500nM) belvarafenib, but not high dose (50nM) cobimetinib. At the transcriptional level, cells treated with the combination showed far greater suppression of negative regulators of MAPK signaling (e.g., DUSPs, SPRY2/4, SPRED1/2) than either agent alone, suggesting potent inhibition of the MAPK pathway by the combination. Intriguingly, OCI-AML3 cells treated with high-dose belvarafenib or low-dose combination unexpectedly exhibited profound down-regulation of mTORC1-regulated genes on GSEA analysis.
We extended these in vitro data by performing preclinical trials in mice transplanted with 5 independent primary Nras- or Kras-mutant AMLs that received belvarafenib (15mg/kg), cobimetinib (2mg/kg), the combination or vehicle by oral gavage (Fig. 2). The combination was well tolerated. Belvarafenib prolonged survival in all 5 trials; in 3 of 5, the addition of low-dose cobimetinib further enhanced survival (median survival: vehicle, 9 days; cobimetinib, 13 days; belvarafenib, 22 days; combination, 32 days; p < 0.0001).
Conclusions
Belvarafenib showed activity in 6 of 6 Ras mutant human AML cell lines and in 5 of 5 primary Ras mutant murine AMLs. Belvarafenib and cobimetinib displayed synergy in all AML cell lines and in 3 of 5 murine AMLs treated in vivo. Mechanistically, we identified distinct biochemical and transcriptional effects of RAF dimer and MEK inhibition in AML cells. We are characterizing these further and pursuing causes of resistance in primary Nras- and Kras-mutant mouse AMLs that relapsed after an initial response to treatment.
Disclosures
Gerosa:Genentech: Current Employment. Dela Cruz:Genentech: Current Employment. Segal:Genentech: Current Employment. Foster:Genentech: Current Employment.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal